See the public tender in your free account here: https://app.hermix.com/opportunities/00195001-2026
A 48-month framework for automatic insulin delivery systems and continuous glucose monitoring across Belgium’s leading university hospitals signals KU Leuven’s commitment to advanced diabetes care for thousands of patients.
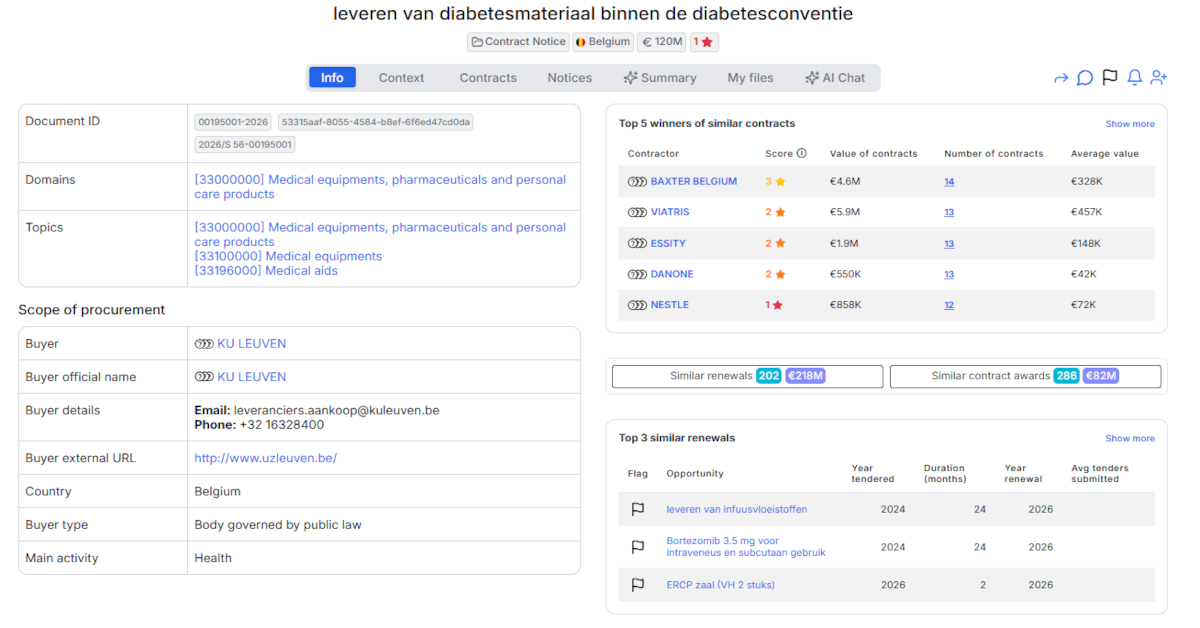
KU Leuven has published a €120 million framework agreement for the supply of diabetes materials under Belgium’s national diabetes convention, positioning this procurement among the largest medical equipment tenders in Belgian higher education and healthcare in 2026.
The framework will supply automatic insulin delivery systems, patch pumps, and real-time glucose monitoring sensors to four hospital networks across the Leuven region: UZ Leuven (the flagship university hospital), AZ Diest, Heilig Hart Leuven, and RZ Tienen. With UZ Leuven alone managing 595 new insulin pump and sensor patients annually, this procurement will directly impact diabetes treatment pathways for thousands of patients over the contract’s four-year lifespan.
Opportunity Overview
Contracting Authority: KU Leuven
Reference Number: PPP0WC-644/6197/P8798 tem P8803
Tender Title: Leveren van diabetesmateriaal binnen de diabetesconventie (Supply of diabetes materials within the diabetes convention)
Scope of Work:
The framework encompasses six distinct lots covering the full spectrum of diabetes technology:
- Automatic insulin delivery pump systems with accessories (Lot 1)
- Automatic insulin delivery patch pump systems with accessories (Lot 2)
- Real-time glucose monitoring sensors for closed-loop insulin systems with accessories (Lot 3)
- Real-time glucose monitoring sensors for convention A patients using stand-alone systems with accessories (Lot 4)
- Real-time glucose monitoring sensors for convention B patients using stand-alone systems with accessories (Lot 5)
- Real-time glucose monitoring sensors for stand-alone system use with accessories (Lot 6)
All medical devices must carry CE marking under applicable legislation and qualify for reimbursement under RIZIV (Belgian National Institute for Health and Disability Insurance) regulations.
Lot Structure:
- Lot 1 (Automatic insulin pump systems): €8,991,396.20
- Lot 2 (Automatic patch pump systems): €8,504,362.24
- Lot 3 (Real-time sensors for closed-loop systems): €6,743,547.16
- Lot 4 (Real-time sensors for convention A stand-alone use): €3,393,811.32
- Lot 5 (Real-time sensors for convention B stand-alone use): €91,787,169.80
- Lot 6 (Real-time sensors for stand-alone systems): €275,471.68
Location: Arr. Leuven, Belgium
Estimated Total Value: €119,695,758.40
Contract Duration: 48 months
Procedure Type: Negotiated with prior publication of call for competition / competitive with negotiation
Award Criteria: Cost and Quality (specific weightings not disclosed)
Key Dates:
- Publication: March 20, 2026
- Submission Deadline: April 21, 2026 (32 days from publication)
Language Requirements: Flemish
EU Funding: No
Financial Guarantee: No security deposit required
Contact Details:
- Email: leveranciers.aankoop@kuleuven.be
- Phone: +32 16328400
Submission Portal: https://www.publicprocurement.be
Ready to qualify tenders like this faster?
Create your free Hermix account at https://hermix.com/sign-up/ and access AI-powered tender analysis, competitive intelligence, and automated monitoring across Europe’s €2 trillion public procurement market.
Authority Profile: KU Leuven and Belgium’s Academic Medical Network
KU Leuven operates as a body governed by public law with health as its primary activity, managing one of Belgium’s most significant university hospital networks. UZ Leuven, the flagship facility, ranks among Europe’s leading academic medical centers for diabetes treatment and endocrinology research.
The authority’s procurement scope extends across four distinct hospital facilities in the Arr. Leuven region, creating a coordinated purchasing network that balances therapeutic freedom for treating physicians with standardized procurement processes. This multi-site framework structure reflects modern Belgian healthcare procurement where academic hospital consortia leverage collective purchasing power while respecting clinical autonomy and patient-specific treatment needs.
Overall Procurement Activity:
- Total Contract Awards: 2,800 contracts worth €1.3 billion
- Active Tender Pipeline: 22 open tenders valued at €21 million
- Renewal Forecast: 1,000 potential renewals worth €1.3 billion
Medical Equipment and Supplies Procurement:
Similar Contract Awards: 285 contracts totaling €81 million
Similar Renewals: 200 upcoming renewals worth €217 million
Other Buyers with Similar Projects: 1,700 authorities representing €2 billion in related procurement
KU Leuven’s procurement history reveals mature medical supply chain management with significant recurring contract cycles. The €1.3 billion renewal pipeline indicates systematic contract renewal practices, while the 285 historical awards for similar medical equipment demonstrate extensive experience procuring diabetes care technologies and related medical devices.
The authority operates within Belgium’s highly regulated healthcare procurement environment where RIZIV reimbursement eligibility drives purchasing decisions and therapeutic freedom principles ensure physicians can select appropriate technologies for individual patient needs.
Competitive Landscape: Pharmaceutical and Medical Device Specialists Dominate
Historical contract awards for similar medical equipment and pharmaceutical supplies reveal a concentrated market led by multinational healthcare corporations and specialized medical device manufacturers.
Winners of Similar Contracts (ranked by total contract value):
- VIATRIS – €5.9M across 13 contracts (€457K average)
- BAXTER BELGIUM – €4.6M across 14 contracts (€328K average)
- GENERAL ELECTRIC – €5.2M across 8 contracts (€739K average)
- ESSITY – €1.9M across 13 contracts (€148K average)
- MEDTRONIC – €1.8M across 4 contracts (€442K average)
- NESTLE – €858K across 12 contracts (€72K average)
- SANDOZ – €832K across 4 contracts (€208K average)
- FRESENIUS – €676K across 11 contracts (€61K average)
- DANONE – €550K across 13 contracts (€42K average)
- KU LEUVEN – 8 contracts (value not disclosed)
Note: Data limited to 10 results; complete contractor list may contain additional suppliers.
Key Competitive Patterns:
Market Concentration: The top three contractors (Viatris, Baxter Belgium, and General Electric) collectively account for €15.7M in contract awards, representing significant market share in KU Leuven’s medical equipment procurement. However, the presence of 10+ active suppliers indicates a reasonably competitive market without complete dominance by any single vendor.
Repeat Winners vs. Specialists: Viatris, Baxter Belgium, Essity, Danone, Nestle, and Fresenius show high contract frequency (11-14 contracts each), suggesting framework agreements or standing arrangements for consumables and ongoing supply needs. In contrast, General Electric and Medtronic demonstrate lower contract frequency but higher average values (€739K and €442K respectively), indicating capital equipment procurement patterns typical of medical imaging, monitoring systems, or advanced therapeutic devices.
Medical Device Specialists: Medtronic’s presence with 4 contracts averaging €442K is particularly relevant, as Medtronic manufactures insulin pump systems and continuous glucose monitoring technologies directly aligned with this tender’s scope. This signals the contractor’s established relationship with KU Leuven for diabetes technology procurement.
Geographic Patterns: The contractor list combines US-headquartered multinationals (General Electric, Baxter, Medtronic), European pharmaceutical companies (Viatris, Sandoz), and specialized medical/nutrition suppliers (Essity, Nestle, Danone, Fresenius). Belgium-based representation appears through Baxter Belgium, indicating local subsidiary operations by international corporations rather than indigenous Belgian medical device manufacturers.
The €81 million in total similar contract awards across 285 contracts yields an average contract value of €284K, significantly lower than this framework’s €120 million total value. This positions the current diabetes materials framework as an exceptional procurement both in absolute value and strategic significance for diabetes care technology deployment across KU Leuven’s hospital network.
Commercial and Procedural Signals
Award Criteria: The tender will evaluate bids on both cost and quality, though specific weightings are not disclosed. For medical devices requiring CE certification and RIZIV reimbursement eligibility, quality factors typically encompass clinical efficacy evidence, patient usability, integration with existing hospital systems, training and support services, and supply chain reliability. Cost evaluation will likely balance unit pricing against total cost of ownership including accessories, consumables, patient education, and technical support.
Procedure Type Implications: The negotiated procedure with prior publication allows KU Leuven to engage in dialogue with bidders after initial submission, creating opportunities to refine technical specifications, clarify RIZIV reimbursement alignment, and negotiate commercial terms. This procedure suits complex medical technology procurement where therapeutic freedom requirements, patient-specific needs, and regulatory compliance necessitate flexibility beyond rigid specification-and-price evaluation.
Language Constraints: Flemish language requirement restricts participation to suppliers with Dutch-speaking commercial and technical support capabilities. While multinational medical device manufacturers typically maintain multilingual operations, smaller specialized suppliers without Benelux market presence may face barriers to participation. This requirement ensures patient education materials, clinical training, and technical documentation align with the Flemish-speaking patient population served by these hospitals.
Lot Structure and Strategy: The six-lot structure enables specialized bidding based on technology categories and patient convention classifications. Suppliers may bid on individual lots matching their product portfolios rather than requiring full-spectrum diabetes technology offerings. The contracting authority explicitly reserves the right to award multiple suppliers per lot to maintain therapeutic freedom and accommodate patient preferences, eliminating exclusive supply arrangements and creating portfolio-based competition.
Lot 5 (real-time sensors for convention B stand-alone use) dominates the framework value at €91.8 million, representing 77% of total contract value. This concentration signals that the majority of diabetes patients at KU Leuven facilities use continuous glucose monitoring in stand-alone configurations under convention B eligibility criteria, rather than integrated closed-loop insulin delivery systems. Bidders with strong sensor technology portfolios addressing convention B patient populations will compete for the framework’s largest value segment.
Contract Duration: The 48-month framework provides supplier stability and patient continuity while allowing technology refresh cycles aligned with medical device innovation timelines. Four-year contracts balance administrative efficiency against the need to incorporate new diabetes technologies as they achieve CE marking and RIZIV reimbursement approval.
RIZIV Reimbursement Dependency: The tender explicitly states that changes in RIZIV reimbursement eligibility at any point during the framework term justify contract termination and new mini-competition procedures. This clause protects KU Leuven from bearing financial risk if devices lose reimbursement status, but creates supplier uncertainty around revenue stability. Bidders must monitor Belgian health insurance policy developments and maintain proactive RIZIV compliance programs to protect contract continuity.
Innovation Reservation: KU Leuven reserves the right to award 90% of volume to primary framework suppliers while holding 10% free for innovative products emerging during the contract term. This provision enables the authority to adopt breakthrough diabetes technologies without full competitive re-procurement, supporting clinical innovation while maintaining framework structure.
Submission Timeline: The 32-day submission window from publication (March 20, 2026) to deadline (April 21, 2026) provides adequate time for bid preparation given the negotiated procedure structure. However, suppliers must rapidly compile CE certification documentation, RIZIV reimbursement evidence, clinical performance data, patient volume estimates, and commercial pricing models across six distinct lots.
No Financial Guarantee: The absence of bid security requirements reduces administrative barriers to participation and signals KU Leuven’s confidence in attracting qualified medical device manufacturers without requiring financial deposits. This approach is typical for framework agreements with established pharmaceutical and medical technology suppliers where corporate reputation and regulatory standing provide implicit assurance.
Strategic Patterns in Belgian Diabetes Care Procurement
KU Leuven’s diabetes materials framework reflects broader structural shifts in European medical technology procurement where academic medical centers consolidate purchasing across affiliated hospitals, leverage RIZIV reimbursement policy as procurement guardrails, and balance therapeutic freedom principles with supply chain standardization.
The €120 million contract value positions this framework among Belgium’s largest single-category medical device procurements in 2026, reflecting the growing diabetes patient population and increasing adoption of continuous glucose monitoring and automated insulin delivery technologies. Lot 5’s €91.8 million allocation for convention B stand-alone sensors indicates that the majority of diabetic patients at KU Leuven facilities qualify for continuous monitoring under broader RIZIV reimbursement criteria rather than the more restrictive closed-loop system eligibility.
Medtronic’s historical presence in KU Leuven procurement with €1.8 million across 4 contracts averaging €442K suggests established vendor relationships for diabetes technology, though the contractor’s relatively lower contract frequency compared to pharmaceutical suppliers (Viatris, Baxter) indicates equipment-based rather than consumables-focused agreements. The competitive landscape data shows no single contractor dominating diabetes device procurement, creating opportunity for both incumbent suppliers to expand market share and new entrants to establish frameworks.
The tender’s therapeutic freedom commitment, multi-supplier award provisions, and 10% innovation reservation collectively signal KU Leuven’s strategy to maintain clinical flexibility while achieving purchasing efficiency. Physicians retain ability to select appropriate technologies for individual patients, while the framework structure standardizes commercial terms, supply logistics, and administrative processes across four hospital sites.
Hermix users analyzing this tender can access competitive intelligence on contractor profiles, authority spending patterns, and renewal forecasting in minutes rather than hours. The platform’s authority profile for KU Leuven reveals 285 similar medical equipment contracts worth €81 million and 200 upcoming renewals valued at €217 million, providing context on procurement cycles, preferred suppliers, and contract timing patterns that inform both bid strategy and account planning for long-term partnerships with Belgium’s leading academic medical network.
The absence of EU funding simplifies compliance requirements while Belgium’s RIZIV reimbursement framework creates a de facto quality and eligibility filter that effectively limits participation to manufacturers with established European regulatory approvals and health insurance recognition.
Practical Takeaways for Bidders
Who this tender suits:
- Medical device manufacturers with CE-marked insulin pump systems, patch pumps, or continuous glucose monitoring sensors currently holding RIZIV reimbursement approval in Belgium
- Suppliers with Flemish-language clinical support, patient education, and technical documentation capabilities serving Dutch-speaking patient populations
- Companies with portfolio breadth across multiple diabetes technology categories enabling multi-lot bidding strategies, or specialized manufacturers targeting specific lots aligned with product strengths
- Vendors with existing relationships or supply agreements with Belgian academic medical centers who can demonstrate experience supporting therapeutic freedom requirements and multi-site hospital network logistics
Critical attention points:
- RIZIV Reimbursement Evidence: Compile current RIZIV approval documentation for all proposed devices, noting that reimbursement status changes during the framework term may trigger contract termination and new mini-competitions
- CE Marking Compliance: Ensure all devices carry valid CE certification under applicable Belgian and EU medical device regulations, with documentation ready for submission
- Therapeutic Freedom Alignment: Structure bids to accommodate physician selection preferences and patient-specific needs rather than exclusive supply commitments, given KU Leuven’s stated commitment to clinical autonomy
- Multi-Lot Strategy: Determine optimal bidding approach across six lots based on product portfolio strength, with particular focus on Lot 5 (€91.8M for convention B stand-alone sensors) representing 77% of framework value
- Innovation Roadmap: Consider how the 10% innovation reservation might enable mid-contract introduction of new technologies achieving CE marking and RIZIV reimbursement during the 48-month framework term
- Patient Volume Estimates: Develop pricing models based on disclosed annual patient volumes: UZ Leuven (595 new patients), AZ Diest (145 patients), Heilig Hart Leuven (64 patients), RZ Tienen (125 patients)
- Submission Deadline: Complete bid preparation, pricing modeling, clinical documentation, and Flemish translation by April 21, 2026 deadline (32 days from March 20 publication)
- Quality Criteria Preparation: Anticipate evaluation criteria beyond price including clinical performance data, patient usability evidence, integration capabilities, training programs, and technical support service levels
- Negotiation Readiness: Prepare for post-submission dialogue typical of negotiated procedures where KU Leuven may seek clarification on RIZIV alignment, technical specifications, or commercial terms
Conclusion
This €120 million diabetes materials framework represents KU Leuven’s strategic commitment to advanced diabetes care technology across Belgium’s leading academic medical network, with the scale and structure to attract Europe’s premier insulin delivery and glucose monitoring manufacturers while maintaining therapeutic freedom and clinical innovation capacity.
The tender’s competitive landscape, multi-lot structure, and RIZIV reimbursement requirements favor established medical device companies with Belgian regulatory approvals and Flemish-language support capabilities, while the negotiated procedure and innovation provisions create pathways for specialized suppliers and breakthrough technologies to enter the framework during its four-year term.
Create your free account today at https://hermix.com/sign-up/ and win more public contracts with the data-driven approach that helps companies consistently succeed in B2G sales across Europe.